Chemical Biology
 Lindsey James, PhD
Lindsey James, PhD
Chemical Biology, Director
ACCEPTING DOCTORAL STUDENTS
The goal of the James lab is to undertake and lead innovative and novel projects focused  on the chemical biology of chromatin regulation, with an emphasis on the development of small molecule chemical probes. Providing such tool compounds to the scientific community has the potential to open new avenues of research in various disease relevant fields and translate to compounds of therapeutic value. Specifically, we are focused on developing compounds to study the domains that recognize the post-translational modification, methylated lysine. Aberrant methylation levels and ensuing changes in gene expression patterns resulting from the altered expression of methyl-lysine (Kme) regulatory proteins is one mechanism by which such epigenetic factors contribute to disease. Kme reader domains have emerged as less precedented epigenetic targets, yet considering the abundant links to cancer genetics, they are well suited to become the next impactful target class of chromatin regulators for intervention.
on the chemical biology of chromatin regulation, with an emphasis on the development of small molecule chemical probes. Providing such tool compounds to the scientific community has the potential to open new avenues of research in various disease relevant fields and translate to compounds of therapeutic value. Specifically, we are focused on developing compounds to study the domains that recognize the post-translational modification, methylated lysine. Aberrant methylation levels and ensuing changes in gene expression patterns resulting from the altered expression of methyl-lysine (Kme) regulatory proteins is one mechanism by which such epigenetic factors contribute to disease. Kme reader domains have emerged as less precedented epigenetic targets, yet considering the abundant links to cancer genetics, they are well suited to become the next impactful target class of chromatin regulators for intervention.
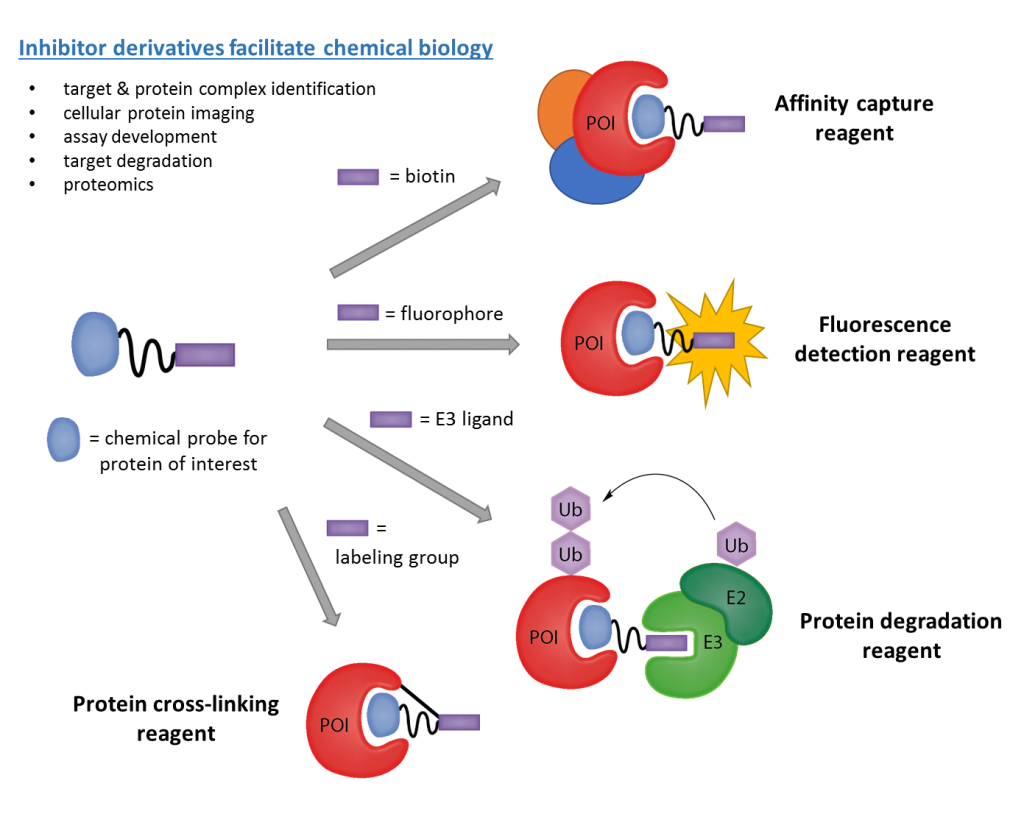
 Our work in this area has pioneered the biochemical assays and medicinal chemistry strategies for high-quality probe development for the methyl-lysine reader target class, as well as the means by which to evaluate probe selectivity, mechanism of action, and cellular activity. Using a variety of approaches, we utilize such chemical tools to improve our understanding of their molecular targets and the broader biological consequences of modulating these targets in disease, particularly cancer. We also think about developing novel methods and screening platforms to discover hit compounds to accelerate Kme reader probe discovery such as affinity-based combinatorial strategies, as well as novel ligand-based tools (see below) such as protein degradation reagents, or PROTACs, as potential therapeutic modalities. We have successfully developed numerous first-in-class chemical probes and currently have compounds in preclinical development (for example, see https://pharmacy.unc.edu/2019/12/pinnacle-hill-announces-first-project-agreement/).
Our work in this area has pioneered the biochemical assays and medicinal chemistry strategies for high-quality probe development for the methyl-lysine reader target class, as well as the means by which to evaluate probe selectivity, mechanism of action, and cellular activity. Using a variety of approaches, we utilize such chemical tools to improve our understanding of their molecular targets and the broader biological consequences of modulating these targets in disease, particularly cancer. We also think about developing novel methods and screening platforms to discover hit compounds to accelerate Kme reader probe discovery such as affinity-based combinatorial strategies, as well as novel ligand-based tools (see below) such as protein degradation reagents, or PROTACs, as potential therapeutic modalities. We have successfully developed numerous first-in-class chemical probes and currently have compounds in preclinical development (for example, see https://pharmacy.unc.edu/2019/12/pinnacle-hill-announces-first-project-agreement/).
Additionally, we collaborate closely with UNC faculty from various disciplines to provide medicinal chemistry and chemical biology expertise to bear on biological targets of therapeutic relevance.
Dr. James is committed to providing an inclusive, safe, and supportive research environment for trainees, teaching trainees to conduct rigorous, ethically sound, and responsible scientific research, and fulfilling the needs of trainees so that they can transition to the next phase of their careers.
Recent Publications
- Ronan P. Hanley; David Y. Nie; John R. Tabor; Fengling Li; Amin Sobh; Chenxi Xu; Natalie K. Barker; David Dilworth; Taraneh Hajian; Elisa Gibson; Magdalena M. Szewczyk; Peter J. Brown; Dalia Barsyte-Lovejoy; Laura E. Herring; Gang Greg Wang; Johnath D. Licht; Masoud Vedadi; Cheryl H. Arrowsmith; Lindsey I. James, “”Discovery of a Potent and Selective Targeted NSD2 Degrader for the Reduction of H3K36me2″” J. Am. Chem. Soc. 2023, March 28
- Frances M. Bashore; Caroline A. Foley; Han Wee Ong; Justin M. Rectenwald; Ronan P. Hanley; Jacqueline L. Norris-Drouin; Stephanie H. Cholensky; Christine A. Mills; Kenneth H. Pearce; Laura E. Herring; Dmitri Kireev; Stephen V. Frye; James, L. I.* “PROTAC Linkerology Leads to an Optimized Bivalent Chemical Degrader of Polycomb Repressive Complex 2 (PRC2) Components” ACS Chem. Biol. 2023, March 6, 2023
- Sun, Z-W.; Waybright, J. M.; Beldar, S.; Chen, L.; Foley, C. A.; Norris-Drouin, J. L.; Lyu, T-J.; Dong, A.; Min, J.;
 Wang, Y-P; James, L. I.*; Wang, Y.* “Cdyl deficiency brakes neuronal excitability and nociception through promoting Kcnb1 transcription in peripheral sensory neurons.” Sci. 2022, 2104317.
Wang, Y-P; James, L. I.*; Wang, Y.* “Cdyl deficiency brakes neuronal excitability and nociception through promoting Kcnb1 transcription in peripheral sensory neurons.” Sci. 2022, 2104317. - Lamb, K. N.; Dishman, S. N.; Waybright, J. M.; Engelberg, I. A.; Rectenwald, J. M.; Norris-Drouin, J. L.; Cholensky, S. H.; Pearce, K. H.; James, L. I.; Frye, S. V.* “Discovery of Potent Peptidomimetic Antagonists for Heterochromatin Protein 1 Family Proteins.” ACS Omega 2022, 7, 716-732.
- Dilworth, D.; Hanley, R. P.; Rerreira de Freitas, R.; Allali-Hassani, A.; Zhou, M.; Mehta, N.; Marunde, M. R.; Ackloo, S.; Machado, R. A. C.; Yazdi, A. K.; Owens, D. D. G.; Vu, V.; Nie, D. Y.; Alqazzaz, M.; Marcon, E.; Li, F.; Chau, I.; Bolotokova, A.; Qin, S.; Lei, M.; Liu, Y.; Szewczyk, M. M.; Dong, A.; Kazemzadeh, S.; Abramyan, T.; Pipova, I. K.; Hall, N. W.; Meiners, M. J.; Cheek, M. A.; Gibson, E.; Kireev, D.; Greenblat, J. F.; Keogh, M-C.; Min, J.; Brown, P. J.; Vedadi, M.; Arrowsmith, C. H.; Barsyte-Lovejoy, D.*; James, L. I.*; Schapira, M.* “A chemical probe targeting the PWWP domain alters NSD2 nucleolar localization.” Chem. Biol. 2022, 18, 56-64.
Recent Funding
4R33DA047023-04 (PI: James) 6/1/2022 – 5/31/2024
NIH, NIDA $1,756,996
Polycomb Repressive Complexes as Key Regulators of HIV Latency and Targets for Latency Reversal
In this proposal we seek to investigate Polycomb regulation of proviral quiescence in HIV infected patients and the role of drugs of abuse in this process in order to advance our current understanding of persistent HIV infection and guide the develop of novel therapeutics, specifically latency reversing agents, toward an HIV cure.
Recent News
January 2023 – Mélanie Uguen was awarded a 2-year postdoctoral fellowship in drug discovery from the PhRMA Foundation – “Multimodal approaches for the develop ment of SETDB1-targeting cancer treatment”
ment of SETDB1-targeting cancer treatment”