 In 2016 a number of scientists, including our own Jeff Aube and Sarah Scarry embarked on a study of small molecules. What they found surprised many.
In 2016 a number of scientists, including our own Jeff Aube and Sarah Scarry embarked on a study of small molecules. What they found surprised many.
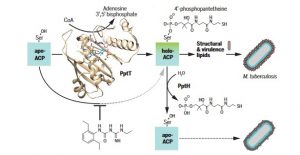
The enzymes PptT and PptH have been found to perform opposing reactions in Mtb lipid metabolism, an essential process demonstrated to be a target for drug development. PptT transfers 4′-phosphopanthetheine (Ppt) from CoA to apo-acyl carrier proteins (Apo-ACP) in Mtb, generating holo-ACPs that help synthesize structural and virulence lipids. Compound 8918 binds to the Ppt binding pocket of PptT, displacing the Ppt arm of CoA and partially inhibiting this enzyme. The Ppt hydrolase PptH can release Ppt, regenerating apo-ACP and thus sensitizing Mtb cells to inhibition of PptT.
Read the full article in Science Magazine here.
